Function and Biogenesis of Cellular Membranes: The Endoplasmic Reticulum
Goal: In this quick and easy lesson, we explain the various metabolic and protein biosynthetic functions of the fantastic endoplasmic reticulum in cell biology.
Learning Objectives
- Define the key metabolic functions of the endoplasmic reticulum.
- Illustrate the functions of the endoplasmic reticulum in the cotranslational translocation of proteins made on membrane-bound polysomes and their co- and post-translational modifications.
- Explain the functions of the endoplasmic reticulum in protein folding and discuss the unfortunate fate of misfolded proteins.
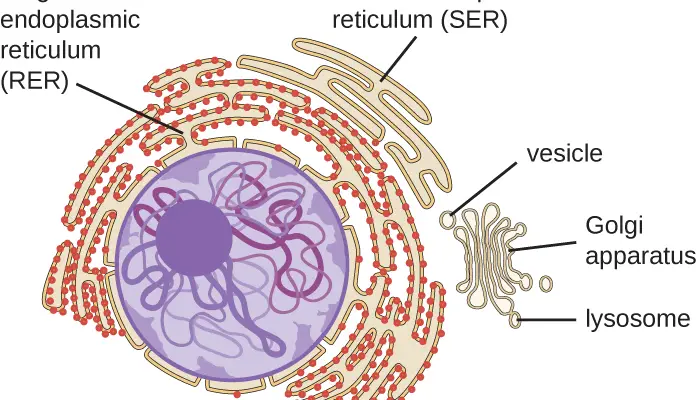
Components of the Endomembrane System (ER)
• Endoplasmic Reticulum (Rough ER, Smooth ER, and Transitional elements, includes outer portion of nuclear envelope)
• Golgi Apparatus (including the cis and trans Golgi Networks)
• Secretory Vesicles and Granules
• Endosomes
• Lysosomes
• Transport Vesicles

Functions of the Endoplasmic Reticulum
• Protein Synthesis and Co- and Post-Translational Modification of Proteins:
–S-S-formation, N-glycosylation, hydroxylation, glycosyl phosphatidyl inositol transfer, polypeptide folding, subunit assembly, quality control and selective degradation of defective proteins.
• Synthesis of Triglycerides, Phospholipids, Cholesterol (and Prostaglandins). De-saturation of fatty acids (by cytochrome b5 and its reductase).
• Calcium Storage and Regulation of Cytoplasmic Calcium Levels.
• Oxidative modification of Low Molecular Weight Products by Cytochromes P450 and
their Reductases.
– hydroxylation of steroids and Vitamin D.
– detoxification of xenobiotics, activation of carcinogens, etc.
• Conjugation of Bilirubin and other Components with Glucuronic Acid.
• Formation of Complexes of MHC Class I Molecules with Immunogenic Peptides.
• Other Specialized Functions, e.g., production of glucose from G-6-P derived
from glycogen, steps in the biosynthesis of steroid hormones and bile acids, etc.

Classes of Proteins Synthesized on the Rough ER (ER Client Proteins)
• Secretory Proteins
• Lysosomal Hydrolases and other Lumenal Proteins of the Endomembrane System
• Integral Membrane Proteins of the Endomembrane System and the Plasma Membrane
(Many of the proteins synthesized in the ER are Glycoproteins)
Components of the Polypeptide Translocation and Folding Apparatus in the Rough ER
Integral membrane Proteins:
• SRP receptor protein
• Sec61 complex/translocation pore
• Signal peptidase
• Oligosaccharide transferase complex (N-linked glycosylation)
Associated Peripheral or Lumenal Proteins
• Chaperone proteins facilitate folding, (include BiP and the glucose binding protein Calnexin)
• Protein disulfide isomerase
Quality control in the ER lumen and preventing ER stress
• Retention of unfolded proteins and components of unassembled complexes by chaperones
• ER stress and its recognition by IRE1, PERK and ATF6 – down regulates protein synthesis (PERK), stimulate mRNA upregulation of more ER protein encoding genes either through mRNA splicing (IRE1=>XBP1) or proteolytic release of transcription factor (ATF6).
• ER Associated Degradation — reverse translocation and degradation via ubiquitinylation of malfolded proteins by cytoplasmic proteasomes
• Autophagy — ER membranes can initiate autophagy through processes controlled by mTOR to degrade accumulated proteins as well as other organelles in cells

ER function and dysfunction in human pathophysiology: Conformational diseases in medicine
• Retention and degradation of mutant proteins may diminish their secretion or expression (Cystic fibrosis, hemophilia, hypercholesterolemia)
• Activation of autophagic processes is essential for turnover of some malfolded proteins to prevent proteotoxicity. Capacity for autophagocytosis may determine the extent of proteotoxicity in an individual with a conformational disease.
• Some malfolded proteins are toxic (e.g., liver disease in 1-antitrypsin deficiency)
• ER stress and its recognition by IRE1, PERK and ATF6 result in upregulation of ER proteins, including chaperones, as well as inhibition of protein synthesis. ER stress can also initiate autophagy through IRE1 and PERK as well as the release of calcium from the ER into the cytoplasm. Chronic ER stress will activate cellular apoptotic pathways though a variety of mechanisms.
Study Questions
- Explain why the lumen of the ER is said to be “topologically equivalent” to the extracellular space.
- Explain why transmembrane glycoproteins of the plasma membrane contain carbohydrate moieties only in their extracellular domains.
- Which of the following organelles belong to the endomembrane system:
- RER - endosomes - nuclear envelope - peroxisomes - phagosomes - mitochondria - trans Golgi network - secretory granules - Discuss the main functions of the ER.
- List the categories of proteins that are synthesized in membrane-bound ribosomes and those that are synthesized in free ribosomes.
- Describe the process of assembly of a membrane bound polysome and the translocation of a secretory protein into the lumen of the endoplasmic reticulum using the following terms. Note that the terms are not in the proper order.
- lumen of the RER - mRNA - nascent polypeptide chain - oligosaccharyl transferase - RER membrane - ribosome - ribosome receptor - signal peptidase - signal sequence - SRP - SRP receptor - translocation pore complex - Define the distinctive characteristic of a:
- primary translation product - insertion signal or signal sequence - proprotein - mature secretory protein - Which co- and posttranslational modifications may a viral envelope glycoprotein undergo before it reaches the plasma membrane?
- What amino acids in a polypeptide chain may serve as acceptor sites for N or O-linked oligosaccharides?
- What is the structure (you do not need to include the stereochemistry of the sugar linkages) of the oligosaccharide chain that is transferred during N-glycosylation to a newly synthesized polypeptide? Which sugar residues may be removed in the endoplasmic reticulum?
- In which organelle of the endomembrane system are the subunits of oligomeric proteins assembled and which proteins facilitate this process?
- What are the mechanisms used to degrade misfolded proteins accumulating in the ER? What is autophagy and what is its role?
- How is the folding capacity of the ER matched to the load of client proteins? What is ER stress? The unfolded protein response?
- Discuss the mechanisms by which mutation(s) that affect the folding of ER client protein might cause human disease.
Click and check out these popular articles for more information: 🙂
Ectoderm vs Endoderm vs Mesoderm
Circulatory System: Blood Flow Pathway Through the Heart
Circulatory System: Heart Structures and Functions
Ductus Arteriosus Vs Ductus Venosus Vs Foramen Ovale: Fetal Heart Circulation
Cardiac Arrhythmias: Definition, Types, Symptoms, and Prevention
Upper Vs Lower Respiratory System: Upper vs Lower Respiratory Tract Infections
Seven General Functions of the Respiratory System
Digestive System Anatomy: Diagram, Organs, Structures, and Functions
Kidney Embryology & Development: Easy Lesson
Psychology 101: Crowd Psychology and The Theory of Gustave Le Bon
Introduction to Evolution: Charles Darwin and Alfred Russel Wallace
Copyright © 2022 Moosmosis Organization: All Rights Reserved
All rights reserved. This essay or any portion thereof may not be reproduced or used in any manner whatsoever
without the express written permission of the publisher.













Excellent essay on this protein making organelle.
LikeLiked by 2 people
Thank you Alex!
LikeLike
great article!
LikeLiked by 2 people
Thank you!
LikeLike
Excellent info. I think there would be a lot of students here at Monash who will appreciate it being passed on ✔
LikeLiked by 2 people
Thank you Barbara!
LikeLike
Excited to hear! Thank you Barbara for your kind comment. Happy learning to the students at Monash! 😀
LikeLiked by 2 people